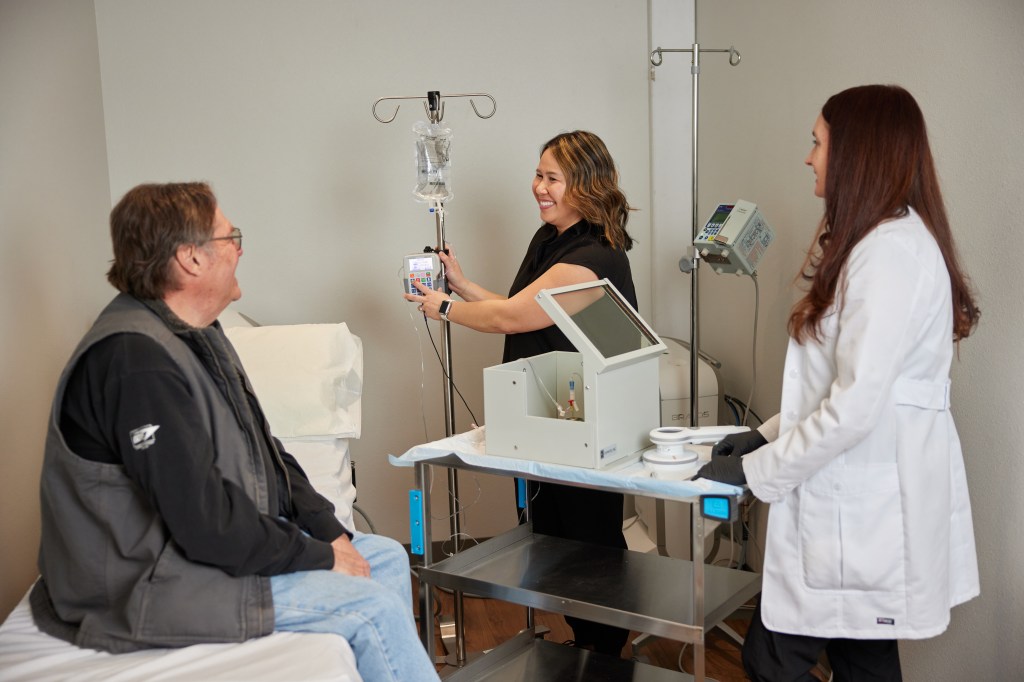
Lutathera (Lutetium-177 dotatate)
We are the first and only site in the state of Nevada to offer this therapy to patients with metastatic midgut neuroendocrine tumors. For patients that are not adequately treated with somatostatin receptor inhibitors alone, Lutathera extends tumor control nearly three years in over half of patients receiving this therapy. Lutathera is a radioactive molecule linked to dotatate, which is injected into the bloodstream and taken up selectively by neuroendocrine cells delivering lethal doses of radiation to the tumor. Injections are given every 8 weeks for a total of four doses in a typical course. A four hour co-infusion of amino acids is given to reduce kidney damage from retention of the Lutathera.
Pluvicto (Lutetium-177 vipivotide tetroxetan)
We are the first and only site in Northern Nevada to provide this therapy to patients with metastatic castrate resistant prostate cancer. This therapy is currently approved as last-line therapy (after disease is not controlled with chemotherapy), but clinical trials are showing promising results to give this therapy earlier for prostate cancer patients. It is a radioactive molecule taken up selectively in prostate cells (and salivary glands). Injections are given every 6 weeks for a total of 6 doses, with most infusions finishing in one hour. Even as last line therapy, the VISION trial showed there is about a 30% imaging response rate, with 9% showing complete resolution of visible disease. Near 50% of patients will show at least a 50% decrease in their blood prostate specific antigen (PSA). Patients receiving this therapy also on average live 4 months longer than prior standard care.